qOMR- Visual Acuity Assessment in Mice
Optomotor Response System
The PhenoSys qOMR (quantitative optomotor response) system is an advanced, automated solution designed to objectively measure visual acuity and contrast sensitivity in mice, enabling precise and reliable assessment of visual function.
qOMR system offers quantitative insights into visual acuity and contrast sensitivity by leveraging the natural optomotor response (OMR).This fully automated system eliminates the need for manual stimulus positioning or specially trained experimenters, making it a robust, simple, and non-invasive method to examine vision in mice.
From characterizing subtle vision defects to tracking disease progression or assessing treatment response, the qOMR offers unparalleled precision and efficiency for your visual research.
The qOMR is a collaboration product brought to market together with its developer, Dr. Friedrich Kretschmer.
https://pubmed.ncbi.nlm.nih.gov/26279344/
https://journals.plos.org/plosone/article?id=10.1371%2Fjournal.pone.0078058

Features
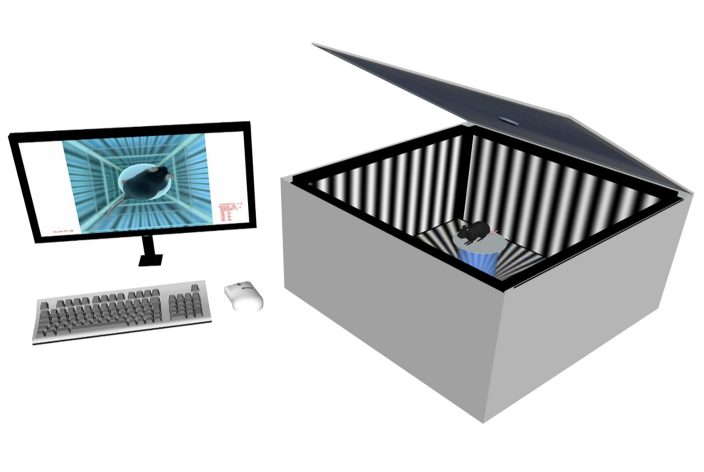
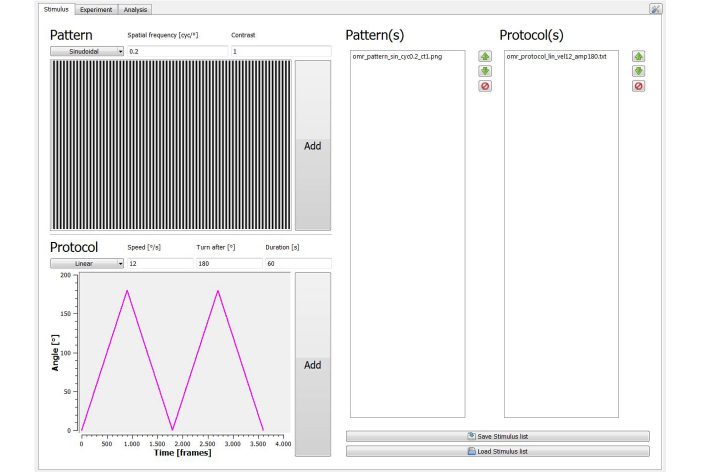
Calibrated 4 screen environment for presenting the virtual stimulation cylinder
Elevated central platform for placing the unrestrained animal. Platform color varies according to the animal's fur color to ensure optimal contrast
Top and bottom mirrors to create the illusion of infinite depth for optimized stimulation
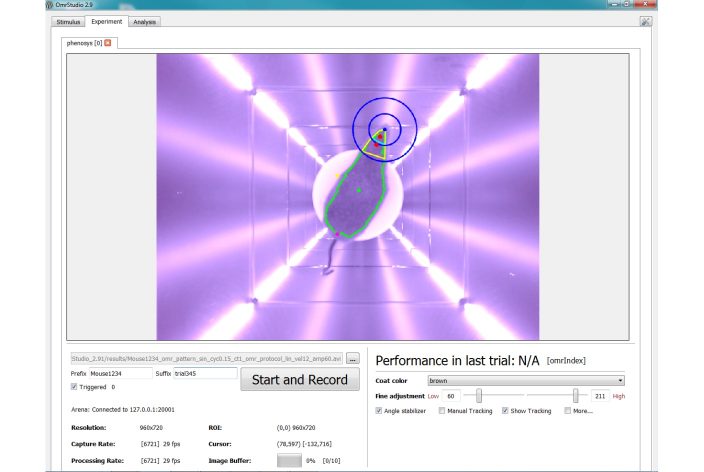
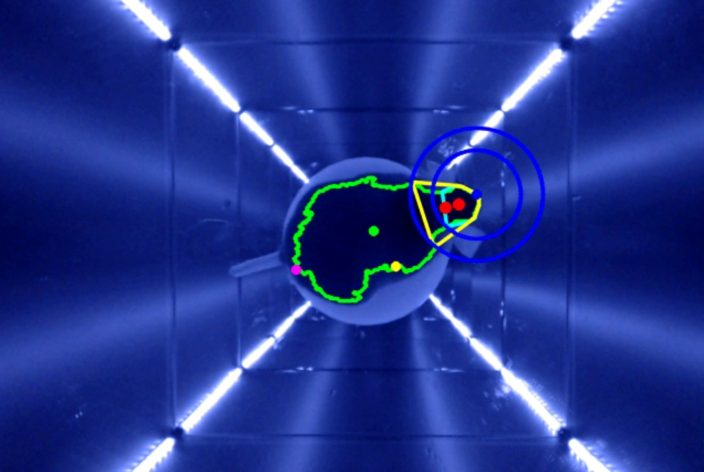
IR-camera for automated head tracking independent of coat color
Adjustable IR-illumination
Anti-reflective (and protective) shields for the screens
Filter set for scotopic measurements
Computer running omrStudio software. Equipped with sufficient computing power for the latest software version.



Key Benefits
Objective & Quantitative Measurements
![]() Eliminates Subjectivity: Provides unbiased, numerical data on visual acuity and contrast sensitivity, bypassing limitations of manual observation.
Eliminates Subjectivity: Provides unbiased, numerical data on visual acuity and contrast sensitivity, bypassing limitations of manual observation.
![]() Reproducible Results: Automated measurement and analysis ensures high consistency across experiments.
Reproducible Results: Automated measurement and analysis ensures high consistency across experiments.
Fully Automated & User-Friendly
![]() Hands-Free Operation: System automatically adjusts stimulus based on real-time head tracking, requiring no manual intervention.
Hands-Free Operation: System automatically adjusts stimulus based on real-time head tracking, requiring no manual intervention.
![]() No Specialized Training Needed: Designed for ease of use, making it practical for a wide range of researchers.
No Specialized Training Needed: Designed for ease of use, making it practical for a wide range of researchers.
Non-Invasive & Animal-Friendly
![]() Stress-Minimized Testing: Assesses vision without physical restraint, sedation, need for surgery promoting natural behavior.
Stress-Minimized Testing: Assesses vision without physical restraint, sedation, need for surgery promoting natural behavior.
![]() Leverages Natural Reflex: Based on the innate optomotor response, eliminating need for animal training.
Leverages Natural Reflex: Based on the innate optomotor response, eliminating need for animal training.
Robust & Reliable Performance
![]() Continuous Alignment: Virtual stimulation cylinder continuously aligns with the animal’s head for accurate data collection.
Continuous Alignment: Virtual stimulation cylinder continuously aligns with the animal’s head for accurate data collection.
![]() Synchronized Head Movement: Precisely evaluates head movements synchronous to stimulation for quantitative OMR.
Synchronized Head Movement: Precisely evaluates head movements synchronous to stimulation for quantitative OMR.
Applications

Characterization of Vision
Objectively measure visual acuity, contrast sensitivity alongwith spectral and temporal sensitivity.

Screening for Vision Defects
Identify and quantify vision impairments.

Tracking Disease Progression & Recovery
Characterization or pre-clinical testing in relevant disease models like glaucoma, or other ocular diseases.

Phenotyping New Breed Lines
Rapidly assess the visual capabilities of novel mouse strains.

Quantification of Treatment Response
Evaluate the efficacy of therapeutic interventions on visual function.
Publications
Featured in numerous peer-reviewed studies across leading neuroscience journals.
Liu Y, Li Q, Yan T, Chen H, Wang J, Wang Y, Yang Y, Xiang L, Chi Z, Ren K, Lin B, Lin G, Li J, Liu Y, Gu F. Adenine base editor-mediated splicing remodeling activates non-canonical splice sites. J Biol Chem. 2023.Large-scale two-photon calcium imaging in freely moving mice
Journal: Journal of Biological Chemistry, 299(12), 105442, 2023
DOI: 10.1016/j.jbc.2023.105442
Kretschmer V, Schneider S, Matthiessen PA, Reichert D, Hotaling N, Glasßer G, Lieberwirth I, Bharti K, De Cegli R, Conte I, Nandrot EF, May-Simera HL. Deletion of IFT20 exclusively in the RPE ablates primary cilia and leads to retinal degeneration. PLoS Biol. 2023.
Journal: PLoS Biology, 21(12), e3002402, 2023
DOI: 10.1371/journal.pbio.3002402
Leinonen, H., Zhang, J., Occelli, L.M. et al. A combination treatment based on drug repurposing demonstrates mutation-agnostic efficacy in pre-clinical retinopathy models. Nat Commun. 2024.
Journal: Nature Communications, 15(1), 5943, 2024
DOI: 10.1038/s41467-024-50033-5
Z Tian, Q Liu, HY Lin, YR Zhu, L Ling, TC Sung et al. Effects of ECM protein-coated surfaces on the generation of retinal pigment epithelium cells differentiated from human pluripotent stem cells. Regenerative Biomaterials, 2024.
Journal: Regenerative Biomaterials, 11(1), rbae091, 2024
DOI: 10.1093/rb/rbae091
Frequently Asked Questions
Customization & Modularity
Can the system be integrated with other systems?
The qOMR is a stand alone product.
Installation and set-up
How long does setup take?
About 2 hours including unpacking.
Is on-site or remote support available?
Yes. Full on-site support, including unpacking, set-up and staff training (one day) is provided.
What are the space/power/environment requirements?
Table 1 m x 1 m.
Data Collection, Storage & Security
How is data collected, stored, and exported?
Data is saved as csv and video files. Plots can be exported for publishing the results easily.
Are backups, encryption, or cloud options available?
Yes.
Technical Support & Troubleshooting
What are the channels which can be used to reach for technical support ?
https://www.phenosys.com/tech-support-form/
Warranty & Service Terms
What’s the warranty period?
One year
What does it cover?
Legally required scope.
Are extended service plans available?
Yes, one year of warranty is included. Each additinal year is charged with 8% of the product price.
Need a quote or consultation?
Contact Us
Get in touch with us and let us know how we can assist you.